- Home
- Guidelines
- Innovation
-
Education
- Study Days & Courses
- STAR Simulation App
- Faculty Resources
- Videos >
- Respiratory Videos (High flow, Tracheostomies, Chest drains, and sleep studies)
- Speciality Training Resources
- Paediatric Long Term Ventilation Team
- Life Support Resources
- #PedsCards Against Humanity
- Bronchiolitis Surge Resources
- Other Educational Opportunities
- Research
- Conference
-
Trainees
- Preceptorships
-
Networks
- Wessex Children's and Young Adults' Palliative Care Network
- PREMIER - Paediatric Regional Emergency Medicine Innovation, Education & Research Network
- Wessex Allergy Network
- Wessex Paediatric Endocrine Network
- Wessex Diabetes Network
- Clinical Ethics >
- TV and Wessex Neonatal ODN
- Regional Referrals to Specialist Services >
- Search
Paediatric Influenza
Introduction
This policy provides guidance on the vaccination of children at risk of severe influenza infection, treatment of presumed/confirmed influenza infection and prophylaxis following significant contact with influenza.
Scope & Purpose
All children at risk of severe influenza, infected with influenza or those in contact with influenza.
Identification of children at risk of severe influenza infection and appropriate immunisation. Appropriate treatment of cases of presumed/confirmed influenza and appropriate management of cases of significant contact with confirmed influenza.
Definitions
Uncomplicated influenza
Influenza presenting with fever, coryza, generalised symptoms (headache, malaise, myalgia, arthralgia) and sometimes gastrointestinal symptoms, but without any features of complicated influenza.
Complicated influenza
Influenza requiring hospital admission and/or with symptoms and signs of lower respiratory tract infection (hypoxaemia, dyspnoea, lung infiltrate), central nervous system involvement and/or a significant exacerbation of an underlying medical condition
This policy provides guidance on the vaccination of children at risk of severe influenza infection, treatment of presumed/confirmed influenza infection and prophylaxis following significant contact with influenza.
Scope & Purpose
All children at risk of severe influenza, infected with influenza or those in contact with influenza.
Identification of children at risk of severe influenza infection and appropriate immunisation. Appropriate treatment of cases of presumed/confirmed influenza and appropriate management of cases of significant contact with confirmed influenza.
Definitions
Uncomplicated influenza
Influenza presenting with fever, coryza, generalised symptoms (headache, malaise, myalgia, arthralgia) and sometimes gastrointestinal symptoms, but without any features of complicated influenza.
Complicated influenza
Influenza requiring hospital admission and/or with symptoms and signs of lower respiratory tract infection (hypoxaemia, dyspnoea, lung infiltrate), central nervous system involvement and/or a significant exacerbation of an underlying medical condition
Guideline
Principles
Treatment
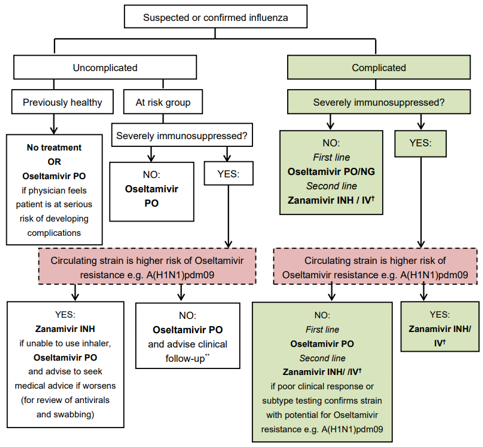
Children with risk factors should be treated with antivirals (as per PHE guidance) if admitted to hospital with confirmed or presumed influenza infection (during periods that influenza is known to be circulating locally).
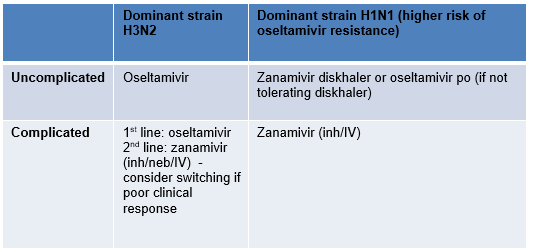
Only treat children without risk factors who are admitted to PHDU / PICU with confirmed or presumed influenza infection. Where indicated, treatment should be initiated as soon as possible (ideally within the first 48 hours of symptoms). The choice of antiviral agent in severely immunocompromised patients depends on the predominant circulating strain H3N2 versus H1N1 (higher risk of oseltamivir resistance with H1N1). See PHE website and briefings during influenza season for further details. If influenza PCR results are subsequently negative in cases of presumed infection, antivirals can be stopped.
Prophylaxis –only children with risk factors should receive prophylaxis following exposure to a person in the same household with confirmed influenza or an influenza-like illness when influenza is circulating in the community. If indicated, prophylaxis should only be used if started within 48 hours of the last contact (use after 48 hours with specialist advice only).
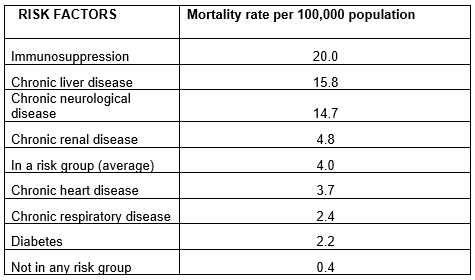
Risk Factors for Severe Infection
- All children with risk factors for severe influenza (see below) should be offered an influenza vaccine (The Green Book - Chapter 19).
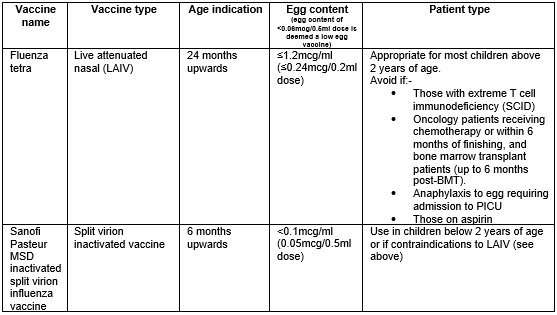
- The indication for vaccination is based on the presence of risk factors for severe infection. The choice of vaccination (live attenuated vaccine (nasal) versus the inactivated vaccine (injected)) is based on the presence of contraindications to the live attenuated vaccine (LAIV). The live attenuated vaccine is cold adapted so is unable to survive above 35°C (so cannot replicate in the lower resp tract) which means that it is safe in the majority of immunocompromised children, including solid organ recipients and children on steroids (≤2mg/kg od). The following groups should receive the inactivated vaccine rather than the LAIV
- Those with extreme T cell immunodeficiency (SCID) or on prednisolone>2mg/kg od.
- Oncology patients receiving chemotherapy or within 6 months of finishing, and bone marrow transplant patients (up to 6 months post-BMT).
- Anaphylaxis to egg requiring admission to PICU
- Patients on aspirin
Treatment
Children with risk factors should be treated with antivirals (as per PHE guidance) if admitted to hospital with confirmed or presumed influenza infection (during periods that influenza is known to be circulating locally).
Only treat children without risk factors who are admitted to PHDU / PICU with confirmed or presumed influenza infection. Where indicated, treatment should be initiated as soon as possible (ideally within the first 48 hours of symptoms). The choice of antiviral agent in severely immunocompromised patients depends on the predominant circulating strain H3N2 versus H1N1 (higher risk of oseltamivir resistance with H1N1). See PHE website and briefings during influenza season for further details. If influenza PCR results are subsequently negative in cases of presumed infection, antivirals can be stopped.
Prophylaxis –only children with risk factors should receive prophylaxis following exposure to a person in the same household with confirmed influenza or an influenza-like illness when influenza is circulating in the community. If indicated, prophylaxis should only be used if started within 48 hours of the last contact (use after 48 hours with specialist advice only).
Risk Factors for Severe Infection
- Under 6 months old from expected delivery date
- Asthma on regular inhaled steroids
- Cystic fibrosis, Primary ciliary dyskinesia or bronchiectasis
- Chronic lung disease on home oxygen or long-term ventilation
- Congenital Heart Disease apart from minor ASD / VSD / PS
- Significant Immune deficiency (any cause including HIV and long term oral steroids)
- Neuromuscular disease e.g. SMA, Duchenne etc
- Significant neurodevelopmental delay
- Other chronic disease where clinician feels decompensation may result from influenza
In general, influenza is less severe in children than in adults. However, children are regarded as super-spreaders of influenza – current DoH recommendations are that all children between 2-15 years should be vaccinated (current uptake of eligible school aged children 51.5% 2021-22)
The eligibility of all children being followed up in general and speciality paediatric out-patient clinics should be assessed and the benefits of immunisation should be communicated to the family. Consider barriers to vaccination (severity of illness, efficacy of vaccine, adverse events) and address in this communication.
Barriers to Vaccination of Children
- Perception of risk of their child getting influenza in terms of likelihood
- Lack of knowledge or misinformation about why healthy children should be vaccinated i.e., risk to other family members/society (population level justification)
- Lack of knowledge about why children with risk factors should be vaccinated re severity of illness
- Practical barriers – getting to GP practice to have the vaccine administered: school vaccination programs being introduced in some areas
- Perception of effectiveness of the vaccine
Side Effects of the Vaccine
- Local pain (10-64%) – related to amount of adjuvant in vaccine (significant in H1N1 pandemic vaccine)
- Other side effects similar to placebo vaccine
- Fever: 3% versus 1%
- Malaise: 9% versus 6%
- Myalgia: 18% versus 10%
Choice of vaccine (LAIV versus inactivated vaccine)
Treatment of influenza
For full treatment guidelines, see here
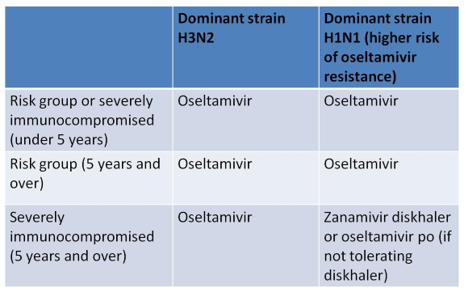
Prescribing Antivirals for treatment of Influenza
Severe immunodeficiency includes:
NOTE: most children with severe immunodeficiency are still eligible to receive the live attenuated vaccine apart from those with extreme T cell immunodeficiency (SCID), oncology patients receiving chemotherapy or within 6 months of finishing or bone marrow transplant patients (up to 6 months post-BMT).
- severe primary immunodeficiency (esp T cell immunodeficiency)
- current or recent (within six months) chemotherapy or radiotherapy for malignancy
- solid organ transplant recipients on immunosuppressive therapy
- bone marrow transplant recipients currently receiving immunosuppressive treatment, or who received it within the last 12 months
- patients with current graft-versus-host disease
- patients currently receiving high dose systemic corticosteroids (equivalent to ≥40 mg prednisolone per day for >1 week in an adult, or ≥ 2mg/kg/day for ≥1 week in a child), and for at least three months after treatment has stopped
- HIV infected patients with severe immunosuppression (CD4<200/μl or <15% of total lymphocytes in an adult or child over five; CD4< 500/μl or <15% of total lymphocytes in a child aged one to five; expert clinical opinion in a child aged under one)
NOTE: most children with severe immunodeficiency are still eligible to receive the live attenuated vaccine apart from those with extreme T cell immunodeficiency (SCID), oncology patients receiving chemotherapy or within 6 months of finishing or bone marrow transplant patients (up to 6 months post-BMT).
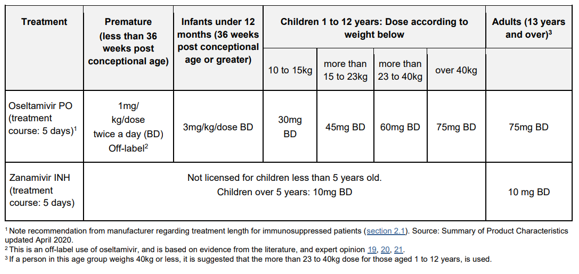
When oseltamivir is indicated based on the above advice, the manufacturer recommends a longer treatment course of 10 days for immunosuppressed patients.
Zanamivir not licensed below age 5 years. Consult paediatric ID team, UHS if use considered in a child below 5 years of age.
The consensus reached by the Wessex network is that children without risk factors (including immunosuppression) with confirmed or suspected influenza should only be treated with oseltamivir if severe enough to require admission to PHDU or PICU. Oseltamivir should be used as standard even for treatment of the immunocompromised child. set. There is evidence that treatment may reduce the risk of mortality even if started up to 5 days after onset. Starting treatment more than 48 hours after onset is an off-label use of oseltamivir and clinical judgement should be exercised. For immunocompromised children with H1N1 being treated with oseltamivir, a follow up respiratory specimen should be collected 48-72 hours after commencing treatment to check that the virus remains sensitive to the drug. Test for antiviral resistance in patients who do not respond after 5 days of treatment.
Prophylaxis following contact with confirmed influenza
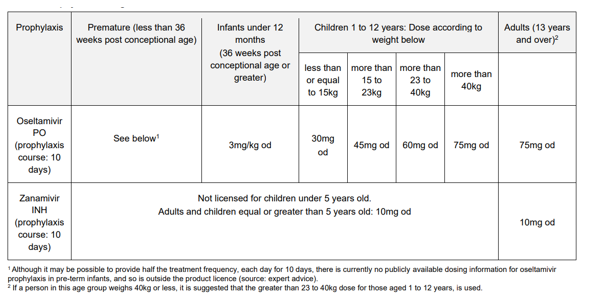
Only children with risk factors should receive prophylaxis following exposure to a person in the same household with confirmed influenza or influenza-like illness when influenza is circulating in the community. If indicated, prophylaxis should only be used if started within 48 hours of the last contact (use after 48 hours with specialist advice only).
Dosing of Antiviral Drugs in Children
Treatment
Prophylaxis
Process for Monitoring Compliance
The purpose of monitoring is to provide assurance that the agreed approach is being followed. This ensures that we get things right for patients, use resources well and protect our reputation. Our monitoring will therefore be proportionate, achievable and deal with specifics that can be assessed or measured.
|
Document Version:
2.0 Lead Authors: Sanjay Patel, Infectious Disease Consultant, UHS |
Approving Network:
Wessex Infectious Disease Network Date of Approval: October 2022 Review Due: October 2025 |
PIER Contact |
|
- Home
- Guidelines
- Innovation
-
Education
- Study Days & Courses
- STAR Simulation App
- Faculty Resources
- Videos >
- Respiratory Videos (High flow, Tracheostomies, Chest drains, and sleep studies)
- Speciality Training Resources
- Paediatric Long Term Ventilation Team
- Life Support Resources
- #PedsCards Against Humanity
- Bronchiolitis Surge Resources
- Other Educational Opportunities
- Research
- Conference
-
Trainees
- Preceptorships
-
Networks
- Wessex Children's and Young Adults' Palliative Care Network
- PREMIER - Paediatric Regional Emergency Medicine Innovation, Education & Research Network
- Wessex Allergy Network
- Wessex Paediatric Endocrine Network
- Wessex Diabetes Network
- Clinical Ethics >
- TV and Wessex Neonatal ODN
- Regional Referrals to Specialist Services >
- Search